
PACK RUST IDENTIFICATION AND MITIGATION STRATEGIES FOR STEEL BRIDGES (Extracts)
Introduction
Corrosion is a major problem in the infrastructure industry, costing millions of dollars every year for maintenance, repair, or replacement. By improving the coating systems and frequency of application, surface corrosion can be effectively controlled. However, very few studies have been conducted to study the influence of pack rust (crevice corrosion) on steel bridges.
When steel elements of a member are unprotected and in contact with another metal, or even non-metal, the steel usually begins to corrode, and rust (iron oxide) starts to develop, or pack in, between the surfaces. Pack rust is not visible until rust product starts deforming the adjoining members and elements. It can cause overstressing of bolts and rivets, and unchecked rust growth may result in bolt and rivet failure. This will reduce the effective capacity of the connection, or might even cause its failure. The Mianus River bridge collapse is an example of the failure of a connection due in part to pack rust formation.
The average age of the existing steel bridges in Indiana is currently about 50 years, and with the continued aging of the bridge infrastructure, the problem of pack rust is most likely going to increase without proactive intervention. In 2012 INDOT included stripe coating in its painting specifications to mitigate pack rust in new structures. But because this adoption is relatively recent, stripe coating for pack rust mitigation is still in question. This study collected quantitative data on the occurrence of pack rust on steel bridges in the state of Indiana and reviewed mitigation strategies used by other DOTs.
Findings
- Pack rust was found to occur frequently in Indiana. About one-third of the state-owned steel bridges exhibit some form of pack rust. The member element most commonly affected (in terms of numbers) is rocker bearings: 318 of 982 bridges showed evidence of pack rust in the rocker bearings. The second member element to frequently exhibit pack rust is a bolted or riveted splice connection: 214 of 1611 bridges were observed to have developed pack rust in a beam or girder splice connection. The members with the highest percentage of pack rust occurrence are gusset plates and hinge-pin connections: pack rust occurred in both of these components in more than 90% of the bridges with such details. End diaphragms, cross bracings, and beam cover plates were also found to be susceptible; however, the frequency of pack rust occurrence in these members is less than 10%.
- The percentage of observed pack rust occurrence for each district was tabulated. Occurrence in the Greenfield and LaPorte Districts is the least among the six districts in Indiana. The LaPorte District, which experiences the highest amount of annual snowfall and also has the highest salt and brine usage in the state, has a pack rust occurrence of 24%, which is less than half of that observed in the Fort Wayne District. There are multiple possible reasons for this observation. One factor that may play a large role is that the LaPorte maintenance crews annually wash the decks and bearings of every bridge using water jets to remove dirt, debris, and salts.
- The occurrence of pack rust in girder and beam splices of bridges that intersect a water body is higher than that of bridges that intersect roads and railroads. The percentage of bridges with pack rust in the splice of exterior beams is higher than that of bridges with pack rust in the splices of interior beams. The study found that it takes 12 years on average after painting a bridge (i.e., re-coating) for crevice corrosion to start in the gap between the members and the splice plates to exhibit visible rust bleeding from the splices. The use of spot painting or recoating at a frequency of less than 12 years may help to minimize pack rust formation. From the point of initiation, it would then take an additional 20 years to reach a very severe pack rust condition.
- With the help of images present in inspection reports, it was observed that the edge distance and the initial pretension in the bolts play a major role in preventing pack rust in splice connections and other connections.
- Stripe coating as a pack rust mitigating strategy is the most popular technique utilized, with 24 state DOTs recommending it in their painting specifications. Thirteen states recommended caulking and 8 states recommended the use of penetrating sealers. Oregon is the only state DOT that outlined a method to repair members affected by pack rust.
- Experimental studies showed that stripe coated connections with the bottom crevice un-caulked experienced the least amount of corrosion and minimum pit depth for new structures. A second series of specimens involved plates that were corroded, cleaned, assembled, and then stripe coated and caulked: caulk placed on all sides was found to produce the best results.
Implementation
- The use of small edge distances with properly tightened high- strength bolts will keep material in firm contact and minimize crevice openings. The use of bolt stagger in new splice connections should be avoided.
- Current INDOT provisions for stripe coating of new structures should be retained. Further study should be done to investigate the effectiveness of stripe coating and the need to modify the number of stripe coats utilized.
- Pack rust formation can be minimized in splice plate details where no pack rust has been detected if the connection region is cleaned and a stripe coat is applied along the crevice at a frequency of no more than 12 years. The opening between the flanges can be sealed with a suitable filler material to prevent moisture entry. If rust bleeding is observed in splice connections, use of an alkaline penetrating sealer appears to be the best option.
- If caulk is used to seal crevices, rust, debris, and salts should be removed and the surfaces cleaned before caulking the crevice. Otherwise they should not be caulked. Caulking an active crevice corrosion cell will likely accelerate the corrosion process.
- Penetrating sealers that are alkaline and have the appropriate viscosity to penetrate into crevices show promising results in mitigating pack rust. The crevice should be cleaned by mechanical tools or high-pressure water jets before applying penetrating sealers. Further study of these sealers should be considered to establish whether they should be used regularly in Indiana.
- Pressurized water jet washing appears to be an effective maintenance practice that reduces the chances of pack rust occurrence in bearings.

HRCSA is a 30 year field proven alkaline penetrating sealer and HRCSA compatible topcoat.
- High Pressure Water Clean (with salt remover) the connection.
Force air dry the connection. - Apply HRCSA penetrating sealer (under pressure) inside connection till refusal. Brush out excess.
- Caulk coat treated connection and surrounding zone with chemically active elastomeric HRCSA topcoat.
- INTRODUCTION
Corrosion is a major problem in the nation’s infra- structure. It is a problem for both structural steel and reinforced concrete structures. Interstate highway construction following World War II generated a sudden demand for bridges to have unobstructed traffic flow. Structural steel was one of the primary materials of choice for bridges in those years.
Figure 1.1 shows the statistics for the number of bridges built in Indiana over several decades. The bridges were designed and built for a service life of about 50–70 years. It is clear that most of the bridges from the 1950s to 1960s are nearing the end of their original intended service life, with many of these bridges needing maintenance and repair. Corrosion is a time dependent process and half a century is a sufficient amount of time for steel to corrode very severely. General corrosion and pitting corrosion are a concern causing section loss and a decrease in load capacity. About 15% of the structurally deficient bridges are deficient because of corrosion (Koch, Brongers, Thompson, Virmani, & Payer, 2002). General and pitting corrosion are visible on the surface. Crevice corrosion, however, is often not visible until it becomes very severe, and can lead to serious problems. Pack rust has been a topic of research interest in the chemical industry. Much research has been conducted to study the behavior of crevice corrosion in stainless steel. The Naval Research Laboratory has conducted some research on iron for crevice corrosion. However, there is limited research being conducted by the bridge industry, although, there is a documented case of a bridge collapse due to pack rust; not directly, but indirectly.
The Mianus River bridge collapse occurred on June 28, 1983 in Greenwich, Connecticut. It was discovered that pack rust displaced the hanger bar of a pin and hanger assembly by 1K inches out of plane from the girder web. This lead to an off-center load on the pin and the ultimate failure was due to the fatigue fracture of the pin see Federal Highway Administration (FHWA, 2015) and National Transportation Safety Board (NTSB, 1984).
- Objective
The study involves three major tasks:
- Literature review:
- Understanding pack rust, or crevice corrosion.
- Understanding how crevice corrosion initiates and propagates.
- Collecting relevant existing research on pack rust.
- Reviewing existing mitigation strategies and repair procedures utilized by other state DOTs.
- Documenting the occurrence of pack rust in Indiana bridges. This task involves answering the following questions:
- Does pack rust occur frequently in the state of Indiana and, if so, is it a problem?
- Which components or members of the bridge are prone to crevice corrosion?
- What parameters influence the formation of pack rust?
- Corrosion
Corrosion is the process of material degradation (both metallic and non-metallic) due to the chemical reaction with the environment. Nature does not store metal in its pure form; it exists in the form of compounds (most common form oxides). Corrosion reactions proceed forward without application of any external energy and metal reaches its stable state. Extraction of metal from its ore needs external energy. Hence, corrosion processes are metal extraction processes in reverse (Fontana, 1987).
- Cost of Corrosion
A study conducted from 1999 to 2001 shows that a total of $276 billion is spent on corrosion-related issues in commercial, residential and transportation sectors. This cost is approximately 3% of the U.S GDP in the year 1998 (U.S GDP in 1998 was $9.09 trillion). The cost of corrosion in the infrastructure industry amounts to $22.6 billion, out of which $8.3 billion is spent on highway bridges. It is observed from the Figure 2.1 the cost of corrosion in highway bridges is about a third of the total cost in the infrastructure industry (Koch et al., 2002).

2.3 Types of Corrosion
Water is often blamed for corrosion. However, corrosion can still be observed in dry conditions where moisture is absent. High-temperature furnace gases can also cause corrosion in steel (Fontana, 1987). This type of corrosion is classified as dry corrosion, and one that occurs due to the presence of water is classified as wet corrosion. Wet corrosion needs an aqueous solution which serves as a path for the ions to flow and com- plete the charge flow circuit.
Different forms of corrosion identified by Jones (1996) are as follows:
1. Uniform corrosion, 2. Galvanic corrosion, 3. Crevice corrosion 4. Pitting corrosion 5. Environmentally induced cracking 6. Hydrogen damage 7. Intergranular corrosion 8. Dealloying 9. Erosion corrosion
Bridges experience all the forms of corrosion from 1 to 5, but hydrogen damage, intergranular corrosion, dealloying corrosion, and erosion-corrosion are not observed in bridges due to the nature of the material used in bridges and the conditions required to cause these forms of corrosion. In this current study, the focus will be on crevice corrosion.
2.4 Crevice Corrosion
Crevice corrosion is the localized form of corrosion which takes place inside the crevice formed by the contact between two metal surfaces or the surface between a metal and non-metal. A portion of the metal which is in contact develops corrosion. Locations on bridge where crevice corrosion is commonly observed include connection details such as splice plates, gusset plates, and the surfaces between bolt/rivet head and
steel plate, and bolt shank and plates. These are the examples of contact between steel and steel. Deposits of sand and dirt on the metal surface can also cause crevice corrosion. The deposits may also act as a shield and the corrosion chemistry depends on the porosity of the deposit.
2.4.1 Mechanism
The fundamental mechanism behind crevice corrosion is still being studied. There are two major theories for the mechanisms of crevice corrosion. The first theory is the traditional theory based on the occluded chemistry change, or the critical crevice solution (CCS), and the second theory is based on the ohmic drop or the IR model. Both the theories are not able to prove all the observations seen in the actual crevice corrosion process.
2.4.1.1 Theory 1. Initially, there is ambient oxygen and aqueous solution present both within and outside the crevice. As the process of uniform corrosion starts, minute local anodic and cathodic sites are formed. The redox reactions taking place inside and outside the crevice are:
Anode: M?Mzn z ne{
Cathode: 2H2O z 4e{ z O2 ? 4OH{
At the start, the kinetics of the reactions are the same throughout as shown in Figure 2.3. The cathodic reaction consumes oxygen. As the corrosion continues, oxygen concentration inside the crevice starts to drop and complete replenishment of the oxygen from the outside is restricted due to the geometry of the crevice. Due to the lack of oxygen in the crevice, the cathodic reaction gets suppressed and the only reaction taking place inside the crevice is the anodic reaction. As the anodic reaction in the crevice increases, the rate of cathodic reaction on the unshielded surface also increases to balance the charge flow and acts as a cathode.
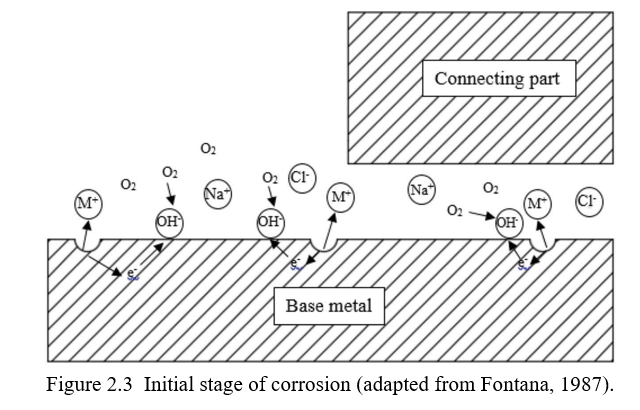
Now that the anodic reaction is localized inside the crevice and the cathodic reaction on the non-shielded surface, the condition is set for the localized corrosion.
With the formation of localized anode and cathode at different locations, a potential gradient is developed between the shielded and non-shielded surfaces. If chlorides are present in the bulk solution, the potential gradient causes the chloride ions to travel from the bulk solution to the crevice solution. Metal ions hydrolyze in the presence of the water and produce hydrogen ions. Increase in the hydrogen ions leads to an acidic crevice solution. Accumulation of the chloride ions and decrease in the pH inside the crevice creates a severe corrosive environment. Due to the formation of the hydroxyl ions in the bulk solution the pH increases, and solution becomes alkaline. The pH of the crevice solution gets stabilized at around pH 3 – pH 4 since hydrolysis is thermodynamically unfavorable below this pH (Pickering & Frankenthal, 1972).
Figure 2.4 shows that as the crevice corrosion first develops the chloride ions start to travel inside the crevice. The concentration of hydrogen ions increases inside the crevice and concentration of hydroxyl ions increases in the bulk solution. The metal ions produced by the anodic reaction starts to move outside the crevice and on their way outside becomes deposited at the mouth

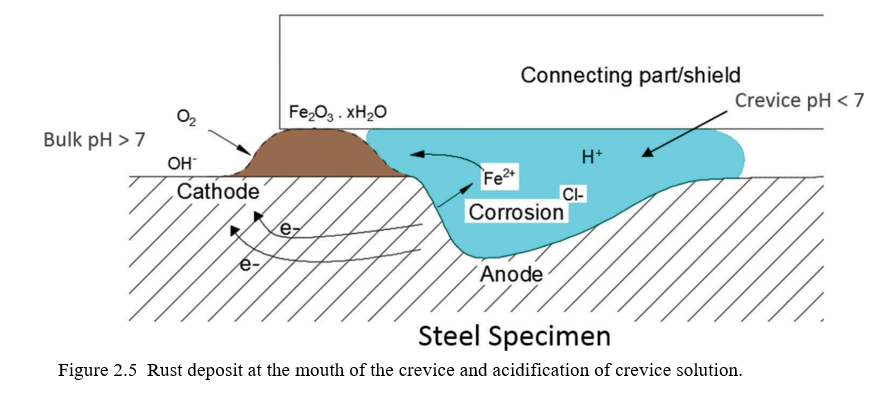
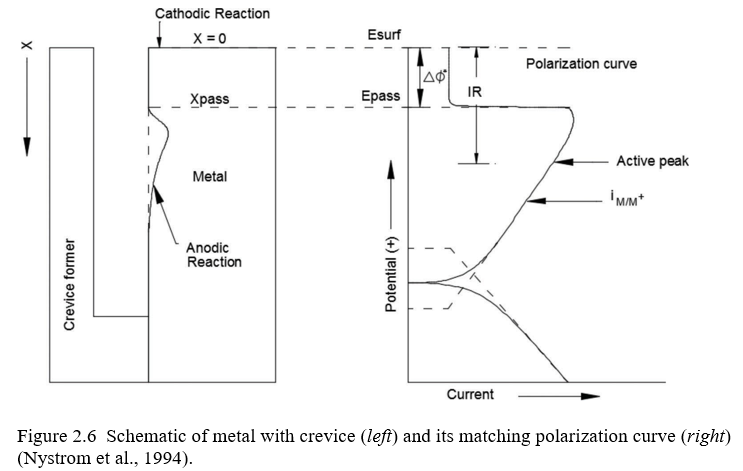
The crevice corrosion process is explained in terms of an electrical circuit, as shown in Figure 2.6. The external surface has a potential Esurf which has a noble potential and thus low dissolution rate. The current, I, flows from the outside surface into the crevice through the aqueous solution of resistance, R, and produces a voltage drop in the crevice. The potential, Ex, at a distance x from the mouth of the crevice can be calcu-lated as by Equation 2.1 (Pickering, 1993):
EX~Esurf {IR ð2:1Þ
Within the crevice, Ex shifts to a less noble potential. The metal surface from the mouth of the crevice to a distance xpass remains passive. At xpass the potential of the metal is Epass, and at this point anodic current peaks to a maximum value. Beyond xpass active dissolution occurs and is known as an active loop. For crevice corrosion to occur the potential drop should be greater than the difference in the potential at the surface and Epass, and is denoted as DW*. Therefore, IR.DW* is the condition for the crevice corrosion to occur (DeForce, 2010). Figure 2.6 on the left shows an experimental setup used at Pennsylvania State University to evaluate crevice corrosion, and on right the crevice polarization curve is depicted. The shape of the corroded region closely resembles the polarization curve (Nystrom, Lee, Sagues, & Pickering, 1994).
Although the IR mechanism can explain the crevice corrosion in a buffered solution (i.e., a constant pH) and in the absence of chlorides, which the traditional mechanism was not able to do, the IR mechanism does not provide a complete understanding of the behavior of crevice corrosion. Further studies are required to find the relation of corrosion kinetics with the size and shape of the crevice. The IR mechanism comes into picture only after crevice corrosion has started. It is not able to explain the initiation process of the crevice
corrosion. This mechanism explains the crevice corrosion behavior in metals which shows passivation during anodic polarization such as stainless steel, titanium and depending on the electrolyte, also iron and carbon steel. Various tests with changing parameters were con- ducted at Pennsylvania State University to observe the behavior of crevice corrosion. The parameters included pH, surface potential, the presence of inhibitors and chlorides in solution, the effect of temperature, crevice
width, and depth.
The experiments at Pennsylvania State University were mainly conducted on stainless steel, which is not the material used in the bridge industry. The experiments were also focused on the environments in the chemical industry. Some experiments conducted on iron were performed on bare specimens and the behavior would be completely different if those were conducted on coated specimens.
- Contradiction in both the mechanism. According to IR mechanism the corrosion is maximum near the mouth of the crevice whereas according to the traditional mechanism, maximum corrosion takes place in the deepest point in the crevice. Clearly, further basic research is needed to understand the mechanics of crevice corrosion, and how it varies for different materials.
- Other Research
The research conducted by Naval Research Laboratory tested some of the parameters involved in crevice corrosion in iron specimens. One of the parameters tested was the crevice height. The relationship of cathodic current and overvoltage by changing crevice height was investigated. The experiments were conducted for 5 mils, 10 mils, 20 mils and 125 mils crevice height. The tests showed that with the decrease in the crevice height
the cathodic current decreases and hence the corrosion rate is reduced. It was seen that the current for the crevice heights in the range 5 to 20 mils remains con- stant. This is thought to be because the thickness of the oxygen diffusion layer in a 0.5N NaCl solution is calculated to be 20 mils (McCafferty, 1974).
The experiments conducted by the Naval Research Board were based on the cathodic polarization, but the experiments conducted at Pennsylvania State University were based on the anodic polarization and thus there will be variation in the results. The variation is because metals do not show passivation in cathodic polarization. To prevent crevice corrosion in bridges carbon steel needs to show passivation behavior with large DW* during anodic polarization. A plasma nitride treatment on carbon steel forms a nitrogen solid solution layer on the surface which increases the corrosion resistance of the carbon steel. The anodic polarization curves show a passivation behavior (Chiba, Nagataki, & Nishimura, 2016). A plasma nitride treatment on the components of the bridge which will be in contact can prevent crevice corrosion. However, this method is expensive and would be very uneconomical at the initial phase for application on bridges. It cannot be recommended without further study on its life cycle cost of maintaining steel bridges over their entire design life.
- MITIGATION AND REPAIR STRATEGIES
- Need for Mitigation and Repair Strategies
It is clear from Figure 1.1 that much of the steel highway bridge infrastructure in Indiana is getting old and has served for a significant percentage of its service life. Figure 2.2 also shows how much money is spent nationally on highway bridges because of corrosion. The highway bridge industry has expended considerable time and resources towards extending the life of bridges by improving the coating (painting) system. The effort was mainly to prevent surface corrosion. However, crevice corrosion was not specifically considered until cases of bolts failure, rivet failure, and excessive plate distor- tions were observed.
Observing the current condition and planning for the future calls for research and development of mitigation strategies for newly built bridges and a pro-active repair procedure for existing bridges. The repair strategies should be able to extend the useful life of existing steel bridge members or elements before they experience a significant strength reduction or total failure.
- Strategies Used by DOTs
Painting specifications of all 50 DOTs were reviewed in search of any provisions specified by the DOT to mitigate pack rust in new bridges or repair procedures for pack rust in existing bridges. Four primary mitiga- ting and repair strategies were found in the coating requirements to tackle pack rust in new and existing bridges. These are as follows:
- Stripe coating 2. Caulking 3. Penetrating sealers 4.Backer rods
- Stripe Coating
A stripe coat is a coat of paint which is applied at the edges, corners, crevices, seams, interior angles, junction of joining members, weld lines or other surface irregularities. The underlying paint coat thickness (primer coat or intermediate coat) at all these locations is less than the paint thickness on the flat surface due to the nature of geometry and the surface tension of the paint film as shown in Figure 3.1. The green film in Figure 3.1 is the paint which is applied to the entire surface, and the blue film is the stripe coat applied on the under- coat system. This additional coat of paint at these locations increases the paint thickness and thus decreases moisture permeability and increases corrosion resistance. The stripe coat at the crevices will significantly increase the paint film thickness and reduce the moisture penetration into the crevice as seen in Figure 3.1. Preventing moisture from entering into the crevice is the motive behind the stripe coat application to prevent crevice corrosion.
The use of a stripe coat was the most common method employed by the DOTs in their effort to mitigate pack rust. There are 24 out of 50 states that require some form of stripe coating in their painting specifications. The paint used for the stripe coat is same as that of the undercoat or the one which will be provided after the application of the stripe coat in some cases. There are multiple application sequences of stripe coat with the painting system used by various DOTs. Some DOTs recommend one stripe coat that is of either primer coat or intermediate coat, while other DOTs recommend up to three stripe coats. In general, it is recommended that the tint of the stripe coat should be in contrast with that of the undercoat, since this makes it easier for the field inspector to inspect the striped location.
The probability of getting cracks in the paint film at the crevices are high if the paint film is too thick. The cracks will also occur due to the differential thermal expansion of adjoining components. Environmental factors and time will also impact the paint and cause cracking. The development of cracks will allow the entry of moisture inside the crevice and promote crevice corrosion. Although there is no particular period established as to when the paint starts to form cracks along the crevices, it is evident that cracks will eventually form. Stripe coat should stay in place and mitigate crevice corrosion until the bridge is repainted after 30 years, which is roughly a typical painting cycle. All this gives an impression that stripe coating cannot serve as a one- time, long-term mitigation strategy but, stripe coating will definitely delay the pack rust formation in steel bridge members.
Pennsylvania Turnpike HRCSA Caulk Coating. No corrosion after 10 years' of HRCSA.

Overview: Around 1998, the bridge was sandblast cleaned and a ‘typically specified’ 3-coat, zinc-basedcoating was applied to the whole bridge, but ZEU did not stop the development of severe corrosionin connections; which became a serious structural problem.
In 2004,the connections between steel plates in the entire bottom chord members had 3” thick pack rust that had bent1” thick steelplates over stressed the rivets.
the crevice corrosion had to be stopped–before the bridge would have to be load posted.
In2006 the Turnpike let a contract that specified zone pressure wash cleaning and application of the Termarust HRCSA coating system.
The project was covered by a 5-yr warranty, with no exclusions.
2016–5 Yr warranty is over–crevice corrosion has been stopped
– no warranty claims

